Haryana State Board HBSE 9th Class Science Solutions Chapter 1 Matter in Our Surroundings Textbook Exercise Questions and Answers.
Haryana Board 9th Class Science Solutions Chapter 1 Matter in Our Surroundings
HBSE 9th Class Science Matter in Our Surroundings Intext Questions and Answers
Questions from Sub-sections 1.1,1.2
Question 1.
Which of the following matter? Chair, air, love, smell, hate, almonds, thought, cold, cold drink, the smell of perfume.
Answer:
Chair, air, almonds, and cold drink matter because they occupy space and mass.
Question 2.
Give reasons for the following observation:
The smell of hot sizzling food reaches you several metres away, but to get the smell of cold food you have to go close.
Answer:
Since, the diffusion becomes faster with the increase in temperature, therefore the smell of hot sizzling? food reaches us from several metres away, whereas to smell the flavour of cold food, we have to go close to them.
![]()
Question 3.
A diver is able to cut through water in a swimming pool? Which property of matter does this observation show?
Answer:
Due to comparatively more distance in between the particles of water, it has the property of compressibility in it, for this reason, a diver able to cut through water in the swimming pool.
Question 4.
What are the characteristics of the particles of matter?
Answer:
The characteristics of the particles of matter are as follows:
- The particles of matter have empty spaces in between them.
- The particles of matter constantly remain in motion i.e., they have kinetic energy in them.
- The kinetic energy of particles of matter increases with an increase in temperature.
- The particles of matter automatically get mixed finally.
- The particles of matter attract one another.
Questions from Sub-section 1.3
Question 1.
The mass per unit volume of a substance is called density. (Density = mass/volume) Arrange the following in order of increasing density – air, exhaust from chimneys, honey, water, chalk, cotton and iron.
Answer:
On writing the given substances in order of increasing density, the following sequence will be obtained:
exhaust from chimneys, air, cotton, chalk, water, honey and iron.
Question 2.
(a) Tabulate the differences in the characteristics of states of matter.
(b) Comment upon the following: rigidity, compressibility, fluidity, Ailing a gas container, shape, kinetic energy and density.
Answer:
(a) Given differences are found in case of different properties of different states of matter:
| Property | Solids | Liquids | Gases |
| Size | They have definite size. | They do not have definite size. | They do not have definite size. |
| Volume | They have definite volume. | They too have definite volume. | They do not have definite volume. |
| Hardness | They are hard. | They are not hard. | They do not have this property. |
| Piling up | They can be piled up. | They flow. | They also flow. |
| Capacity of compression | They cannot be compressed. | They cannot be compressed. | They can be compressed. |
| Position of Particles | Particles are very close with each other. | Particles are away from each other. | They have too much vacant space in between particles. |
(b) (i) Rigidity: The force of cohesion working in between the particles of a substance determines the rigidity of the substance. Due to the maximum force of cohesion in solids, rigidity is there. Liquids have yet lesser and the gasses have the least rigidity.
(ii) Compressibility: Reducing of interstitial gaps in the particles of a substance with the application of external force is called compressibility. Gases have the property of compressibility.
![]()
(iii) Fluidity: Materials that have the property to flow are known as liquid materials. In liquids, the property of fluidity is there.
(iv) Filling of gas Container: Due to weak intermolecular force between the gas particles in a container, the gas occupies the whole of the provided space, which means gases do not have a definite volume. The gases are filled in the container at high pressure.
(v) Shape: Since the particles of solids are attached to more intermolecular force, hence they provide a definite shape to the solids. Whereas, in liquids the intermolecular force is comparatively low, due to this reason liquids do not have a definite shape. The same is the case with gases too.
(vi) Kinetic Energy: The energy produced due to the speed of particles is called kinetic energy. On increasing the temperature the kinetic energy of particles also increases. There is not much kinetic energy in solids, some kinetic energy in liquids whereas gases have high kinetic energy. Density of soilds is high, low in liquids whereas it is nil in gases.
(vii) Density: The mass per unit volume of a substance is called density, i.e.,

Question 3.
Give reasons:
(a) A gas fills completely the vessel in which it is kept.
(b) A gas exerts pressure on the walls of the container.
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood we need a karate expert
Answer:
(a) As the volume of gas is not definite, the gas completely fills up die vessel in which it is kept.
![]()
(b) In the gaseous state the speed of particles is irregular and it is fairly random. Due to this irregular speed, the particles of gas collide with themselves and with the walls of the container. Due to the cohesive force exerted by die particles of gas per unit area; on the walls of the container, the pressure of the gas is maintained.
(c) As the wooden table constitutes a definite shape, also it has a definite volume and the property of incompressibility; therefore, it is called to be solid.
(d) Due to maximising space in the particles of air, we can move our hand quite easily in the air, whereas, due to less intermolecular space between the particles in the piece of wood, we have to be quite expert at Karate Technique.
Question 4.
Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why.
Answer:
In comparison to solids, liquids have low density, but water has maximum density at 4°C. When it is cooled down below 4°C, the ice starts thawing at 0°C which has lower density than the density of water. Therefore ice cube floats on the surface of water.
Questions from Sub-section 1.4
Question 1.
Convert the following temperatures to Celsius scale:
(a) 300 K
(b) 573 K.
Answer:
(a) 300 K = (300 – 273)°C = 27°C
(b) 573 K = (573 -273)°C = 300°C
Question 2.
What is the physical state of water at:
(a) 250°C
(b) 100°C?
Answer:
(a) At 250°C water remains boiling.
(b) At 100°C water will start boiling, because this is the boiling point of water.
![]()
Question 3.
For any substance, why does the temperature remain constant during the change of state?
Answer:
During the change of state in any material the temperature remains constant because during the change of state the entire heat energy being yielded to the material brings under control intercohesive force of the particles, which is utilised to change the state of a material.
Question 4.
Suggest a method to liquefy atmospheric gases.
Answer:
The atmospheric gases present in the air zone are confined to a utensil and by increasing its pressure and reducing the temperature, they can be converted further into liquid.
Questions from Sub-section 1.5
Question 1.
Why does a desert cooler cool better on a hot dry day?
Answer:
On a hot and dry day, due to lack of humidity, the air is generally dry, when this dry air is sucked inwards by the exhaust fan of the desert cooler, then the water trickling on the mats of the desert cooler turns into vapours and thereby sucks the heat present in the dry air, as a result, the air around cools down. This cool air cools down the room from the inside.
Question 2.
How does the water kept in an earthen pot (matka) become cool during summer?
Answer:
The earthen pot consists of very small pores in its walls through which the water leaks out from inside the pot with capillarity process. These small drops of water get evaporated and it receives heat energy from water itself for evaporation. Consequently, the water kept in the earthen pot remains cold.
![]()
Question 3.
Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Answer:
By pouring down acetone/petrol or perfume on the palm, its particles eliminate heat energy from the palm and further, evaporate it into air due to which the palm feels cold.
Question 4.
Why are we able to sip hot tea or milk faster from a saucer rather than a cup?
Answer:
The surface area of the saucer is far more than that of a cup due to which, the evaporation in case of saucer is more rapid from its surface and with that the hot milk or tea comparatively cools down quicker and due to their cooling down, they can be drunk quickly.
Question 5.
What type of clothes should we wear in summer?
Answer:
In summer, we should wear cotton clothes, it is because of the reason that in summer, due to physical activity we perspire more, with a result we feel cool. As we know during evaporation the particles of the liquid’s surface receive energy from our body and further convert it into vapours.
The exposed dormant heat energy of evaporation combines with the equal amount of heat energy absorbed from our body, which lets our body feel cool. By wearing cotton garments the absorption of water gets more, consequently the sweat after absorption into it gets easily evaporated into the atmosphere.
HBSE 9th Class Science Matter in Our Surroundings Textbook Questions and Answers
Question 1.
Convert the following temperatures to the Celsius scale:
(a) 293 K
(b) 4701L
Answer:
(a) 293K = (293 – 273)°C = 20°C
(b) 470K = (470-273)°C = 197°C
Question 2.
Convert the following temperatures to the Kelvin scale:
(a) 25°C
(b) 373°C.
Answer:
(a) 25°C = (25 ÷ 273)K = 298 K
(b) 373°C = (373 + 273)K = 646 K
Question 3.
Give the reason for the following observations.
(a) Naphthalene balls disappear with time without leaving any solid.
(b) We can get the smell of perfume sitting several metres away.
Answer:
(a)Naphthalene ball is after some time without losing its solid state disappear, it is because of the reason that naphthalene is a volatile substance which directly converts from a solid state into a gaseous state.
(b) We can feel the fragrance of perfume sitting several metres away because perfume has the property to diffuse into air.
![]()
Question 4.
Arrange the following substances in increasing order of forces of attraction between the particles: water, sugar, and oxygen.
Answer:
In increasing order the applicable intermolecular force among the molecules will be as follows:
Oxygen < Water < Sugar.
Question 5.
What is the physical state of water:
(a) 25°C,
(b) 0°C, (c) 100°C?
Answer:
(a) At 25°C water will be in a liquid state.
(b) At 0°C water will be in a solid state (ice).
(c) At 100°C water will be in a boiling state (steam).
Question 6.
Give two reasons to justify:
(a) water at room temperature is a liquid.
(b) an iron almirah is solid at room temperature.
Answer:
(a) Water at room temperature is in a liquid state, because
- At this temperature the applied force of cohesion between its molecules used to be normal.
- Its molecules have normal elliptical speed.
(b) The iron almirah at room temperature is in solid state, because
- The applicable force of cohesion in between the molecules of iron is fairly excessive.
- The gap distance between the molecules of iron is almost negligible due to that reason they can move around at a fixed distance.
Question 7.
Why is ice at 273 K more effective in cooling than water at the same temperature?
Answer:
At 273 K temperature, in comparison to water, ice releases more coolness, because in ice inexposed melting heat energy is more.
![]()
Question 8.
What produces more severe burns, boiling water or steam?
Answer:
Rather, steam will give severe burning sensation it is because of the reasons that steam does have extra heat energy in it which is called in exposed heat energy of evaporation.
Question 9.
Name A, B, C, D, E and F in the following diagram showing changes in its state:
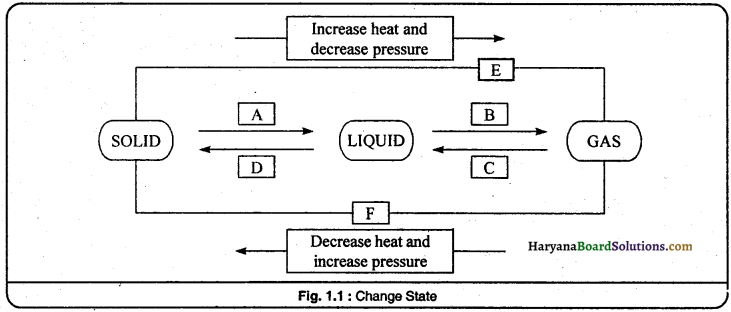
Answer:
(A) Melting
(B) Vaporisation
(C) Condensation
(D) Freezing
(E) Sublimation
(F) Sublima