Haryana State Board HBSE 6th Class Science Solutions Chapter 5 Separation of Substances Textbook Exercise Questions and Answers.
Haryana Board 6th Class Science Solutions Chapter 5 Separation of Substances
HBSE 6th Class Science Separation of Substances Textbook Questions and Answers
Exercises
Question 1.
Why do we need to separate different substances present in a mixture? Give two examples.
Answer:
We need to separate different substances present in a mixture due to the following purposes:
- To remove the unuseful or harmful components for example removing tea leaves after making tea.
- To obtain the useful components – for getting wheat or rice grains after separating.
- To remove impurities for getting a pure sample. For example obtaining sugar crystals from jaggery (Gur) or salt from sea water.
Examples:
- Tea leaves age separated from liquid with a strainer while preparing tea.
- Grain is separated from stalks, while harvesting.
Question 2.
What is winnowing? Where is it used?
Answer:
The process of separating lighter and heavier particles from a mixture is called winnowing. In this process/method heavier particles of grains are separated from lighter particles of husk. The husk particles are carried away by wind and separate heap away from that of the heavier particles of grains of wheat is formed. This method is used by farmers in fields as well as at a flour mill (atta chakki).
Question 3.
How will you separate husk or dirt particles from pulses before cooking?
Answer:
The husk particles or dust particles are separated from pulses by the method of hand picking. Since the undesired particles are present in small quantities and the total amount of pulses to be separated is generally not very- very large. So they are separated by hand picking before cooking the pulses.
![]()
Question 4.
What is sieving? Where it is used?
Answer:
Sieving is a method used to separate the components of a mixture which are of different size. The undesirable particles in the form of husk remain on the seive and fine particles of flour pass through the holes of the sieve. Sieving flour before consuming is not a good practice, because during sieving husk remains on the seive, which provides us roughage which is useful for our body.
Question 5.
How will you seperate sand and water from their mixture?
Answer:
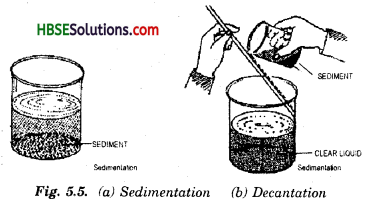
A mixture of sand and water can be separated by decantation method. Take a glass tumbler half filled with the given sample of water. Stir the water thoroughly. Now keep the tumbler undisturbed for sometime. We must see that sand being heavier than water has settled down at the bottom of the tumbler. This process is sedimentation. Now pour the. clear water into another tumbler, without disturbing the layer of sand and with the help of a glass rod. This process is decantation. In this way, mixture of sand and water can be separated.

Question 6.
Is it possible to separate sugar mixed with wheat flour? If yes, how will you do it?
Answer:
Yes, it is possible to separate sugar mixed with wheat flour. The mixture of sugar & wheat flour can be separated by sieving. The wheat fine particles separates by strainer and sugar particles being bigger in size remains on the strainer. Thus sugar can be separated from the mixture of sugar and wheat flour.
Question 7.
How would you obtain clear water from a sample of muddy water?
Answer:
We can separate clear water from muddy water by loading process. Take a beaker half filled with muddy water and take a piece of alum. Tie the alum piece by a cotton thread.
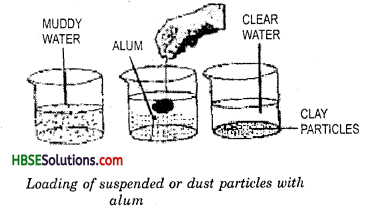
Now dip the alum in water and move it slowly in a circular path for about one minute. Remove the alum and keep the beaker undisturbed for a few minutes. We find that dust particles that are responsible for making the water muddy settle down readily. Alum dissolves in water very easily. The fine dust particles get loaded and become heavier and settle down at the bottom of the beaker. The clear water may be decanted. This activity combines two methods loading and decantation.
![]()
Question 8.
Fill up the blanks :
(a) The method, of separating seeds of paddy from its stalk is called ________.
(b) When milk is poured onto a piece of cloth the cream(malai) is left behind on it. This process of separating cream from milk is an example of ________.
(c) Salt is obtained from sea water by the process of ________.
(d) Impurities settled at the bottom when muddy water was kept over night in a bucket. The clear water was then poured of from the top. The process of separation used in this example is called ________
Answer:
(a) Winnowing
(b) filtration
(c) Evaporation
(d) Decantation.
Question 9.
True or False?
(a) A mixture of milk and water can be separated by filtration.
(b) A mixture of powdered salt and sugar can be separated by the proces of winnowing.
(c) Separation of sugar from tea can be done with filtration.
(d) Grain and husk can be separated with ‘ the process of decantation.
Answer:
(a) False
(b) False
(c) False
(d)True.
Question 10.
Lemonade is prepared by mixing lemon juice and sugar in water. You wish j to ice to cool it. Should you add ice to the lemonade before or after dissolving sugar? In which cash would it be possible to dissolve more sugar?
Answer:
We should add ice in lemonade after dissolving sugar in it, because sugar dissolve more before adding ice. It would be possible to dissolve more sugar before adding ice in the lemonade, because it dissolve more into hot than in cold.
HBSE 6th Class Science Separation of Substances Important Questions and Answers
Very Short Answer Type Questions
Question 1.
What is a pure substance?
Answer:
A pure substance is one that consists of a single element or compound.
Question 2.
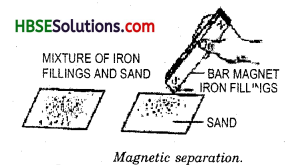
How will you separate a mixture of iron fillings and sulphur powder?
Answer:
The mixture can be separated by using a magnet. The iron fillings are picked out by the magnet and sulphur powder remains behind.
Question 3.
Name the method used to separate butter from milk.
Answer:
Centrifugation/churning method.
![]()
Question 4.
Name two materials used as filters.
Answer:
- Cotton cloth
- Filter paper.
Question 5.
Which method will you use to separate blue vitriol (Neela thotha) from its solution?
Answer:
Crystalization method.
Question 6.
What is meant by the term ‘mixture’?
Answer:
A mixture is a substance made up of two or more elements or compounds, physically mixed in any ratio.
Question 7.
Give three examples of mixture.
Answer:
Tea, milk and air.
Question 8.
Write two properties of mixtures.
Answer:
- Ratio of constituents is not fixed.
- Does not have definite m.p. or b.p.
Question 9.
Name the process of obtaining ; iron from waste materials.
Answer:
Magnetic separation.
Question 10.
How can you obtain distilled water?
Answer:
By distillation process.
Question 11.
Name the process to separate two immiscible liquids.
Answer:
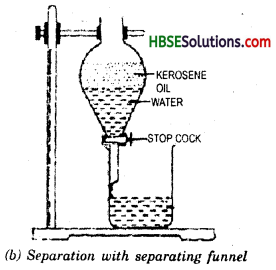
Decantation process by separating funnel.
Question 12.
Name a gaseous mixture.
Answer:
Air.
![]()
Question 13.
Name three mixtures found in nature.
Answer:
- Sea water/pond water/lake water.
- Air
Question 14.
What is winnowing?
Answer:
It is the process of separation of grains from husk and hay with the help of wind.
Question 15.
What is centrifugation?
Answer:
It is a method of separating suspended solid particles from a liquid.
Question 16.
Name the method of separating two immiscible liquids.
Answer:
Decantation or By separating funnel.
Question 17.
What is sublimation?
Answer:
It is a process in which a solid gets converted into its gaseous state.
Question 18.
What is loading?
Answer:
The process of setling down solid particles quickly.
Question 19.
What is churning?
Answer:
Churning is a method of making butter.
Short Answer Type Questions
Question 1.
What is a mixture? Give two examples.
Answer:
A mixture is a substance made up of two or more elements or compounds, when physically mixed in any ratio.
(i) Gun powder (ii) Salt in water.
Question 2.
How will you prepare a big crystal of sugar from powdered sugar?
Answer:
Prepare sugar solution in hot water and allow it to cool. Filter the solution. Now hang a crystal of sugar with the help of a thread and a glass rod leave the solution undisturbed for four days. Now we see that crystals slowly begin to grow on the crystal.
![]()
Question 3.
How is common salt obtained from sea water?
Answer:
Sea water is run into shadow ponds and allowed to evaporate to dryness in hot sun. Water evaporates and solid common salt is left behind. The salt is then collected in heaps.
Question 4.
How will you separate a mixture of sulphur in water?
Answer:
Take the mixture of water and sulphur in a beaker. Pour the mixture on the filter paper with the help of a glass rod so that the liquid stands below the edge of the cone. The residue that remains on the filter paper is sulphur. Now dry it to get sulphur and water is collected in beaker.
Question 5.
How will you separate a mixture of naphthalene and salt?
Answer:
Naphthalene and salt mixture can be separated by the process of sublimation. Naphthalene sublimates and is collected on inverted keep and salt remains behind in the dish.
Question 6.
What do you mean by decantation? What are its uses?
Answer:
Decantation is a process by which we can remove a liquid without disturbing the solid settled down in the bottom.
Uses: This method can be used to separate a mixture of
- sand and water
- mixture of two liquids – water and oil.
![]()
Question 7.
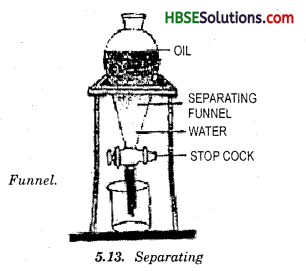
How will you separate a mixture of water and kerosene oil?
Answer:
Mixture of kerosene oil and water can be separated by using separating funnel. Take the mixture in separating funnel and allow it to stand for some time. After sometime, two different layers form. Kerosene oil being lighter forms upper layer and water forms lower layer. By opening the stop cock, we obtain water and kerosene remains on the funnel.
Question 8.
What do you mean by distillation? How can you separate a mixture of alcohol and water?
Answer:
Distillation is the process in which liquid is heated to get vapours and then cooling the various to get back liquids. It is used to separate a mixture the components of which have different boiling points.
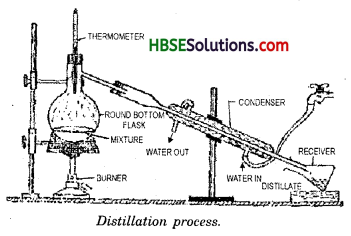
The mixture of alcohol and water is taken in a round bottom flask. Alcohol boiling point 80°C, evaporates first on heating as compared to water with boiling point 100°C. Now alcohol vapours are condensed by cooling in a separate container. It gets converted back to alcohol liquid and water remains in flask.
Question 9.
How will you separate a mixture of iron dust and sulphur powder?
Answer:
A mixture of iron dust and sulphur can be separated by the magnetic separation method. Iron dust is attracted towards the magnet and separated in a container and sulphur remains behind in the container.
Question 10.
How will you separate a mixture of iodine and sand?
Answer:
A mixture of iodine and sand can be separated by sublimation. The mixture is taken into a china dish and covered with inverted funnel. A cotton plug prevents the vapours from escaping into atmosphere. On heating, iodine sublime and collected on the cool inner surface of funnel. Sand is left behind in the disc.
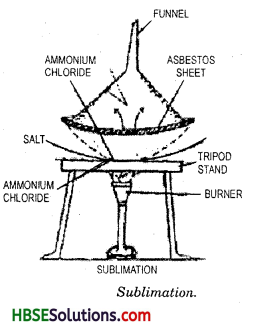
Long Answer Type Questions
Question 1.
How will you separate a mixture of iron fillings, common salt, sand and sulphur?
Answer:
Iron fillings are separated from the mixture by using a magnet. The remaining mixture of salt,-sand and sulphur is stirred with water when salt is dissolved in it. On filtration salt solution is obtained as a filtrate and the residue contains sand and sulphur.
Sand and sulphur mixture is shaken with carbon disulphide, when sulphur dissolves in it but sand remains undissolved. On filtration, sand is obtained as residue and sulphur as filtrate. On evaporating the filtrate carbon disulphide is removed and sulphur is obtained.
![]()
Question 2.
How you will purify common salt obtained from sea water?
Answer:
The lumps of impure common salt obtained from sea water, are crushed to get powdered salt. The powdered common salt is dissolved in water to prepare a solution. The solution of common salt is filtered to remove insoluble impurities.
The clear solution of common salt is evaporated by heating to remove some of the water to obtain a concentrated solution. The hot concentrated solution of common salt is allowed to cool. On cooling, the hot solution crystallises and crystals of pure common salt are obtained.
Separation of Substances Class 6 HBSE Note
1. We use some materials like sugar, common salt, water, milk as such in our daily life. In many materials, we have to separate the useful components from the unwanted or sometimes harmful components before we can use them. For example we peel off the skins of mangoes and bananas before using them.
2. Pure substances : A pure substance is one that consists of a single element or compound. All its constituents have similar properties. Examples are copper, iron, water, common salt, sugar etc.
3. Mixtures : A mixture contains two or more types of constituents which are present in any ratio. For example, air around us contains many gases such as nitrogen, oxygen, carbon dioxide and dust particles. Thus air is a mixture. Sea water, pond and river water are also mixtures. Gun powder and brass are other examples of mixtures.
4. Need for separation : We know that a mixture contains two or more components. We want to be very sure about the purity of the food we take in. Even the water we drink should be pure. We can say that we carry out the separation of substances with the following purposes:
- To remove the un-useful or harmful component.
- To obtain the useful component.
- To remove impurities for getting a pure sample.
5. Principle of separation : Different materials have different properties and we can use these property differences to separate mixtures. The components of a mixture can be separated by one method or by a combination of various methods.
6. Method of separation : Often, in mixtures, we are able to see the different components present. There are several methods used for separating the constituents of a mixture. The choice of the method used depends upon the nature or the properties of the constituents. Some common methods for the separation of the components of a mixture are as follows :
7. Hand Picking: This method is use for separating small particles of dust, stone, husk etc. from the grains of wheat, rice and pulses etc. Since these unwanted components are present in small quantities and total amount of grains to be separated is not very-very large, so they are separated by hand picking.

8. Sieving: Sieving of wheat flour (atta) is a common practice. Sieving a wheat flour through a sieve allows fine particles of the flour to pass through the holes of the sieve while the bigger husk particles remain on the sieve.

Sieving is used when two components of a mixture have different sizes. Sand can be separated from stone by seiving.
9. Winnowing : This method of separation is commonly used to separate lighter particles from heavier grains of wheat.

In this method, the wheat grains containing husk are allowed to fall down from some height while the wind is blowing. Since husk is lighter than grains, it gets carried away by wind and heaps of grains and husk are formed separately.
10. Sedimentation and decantation : This method of separation is used for a mixture consisting of an insoluble solid and a liquid. The process of settling down of heavier insoluble particles in a mixture of water and insoluble substances is called sedimentation (Fig 5.5). The process of transferring the clear liquid (after sedimentation) without disturbing the sedimentation insoluble heavy particles is called decantation. Fig. 5.5. (6).
Decantation can also be used for separating two immiscible liquids such as oil and mixture. Oil and water do not mix with each other and form separate layers. They can also be separated by using separating funnel.
11. Loading : In rainy season we get muddy water from wells, taps or other sources of drinking water. This muddy water can be made clear by loading. If a piece of alum, tied
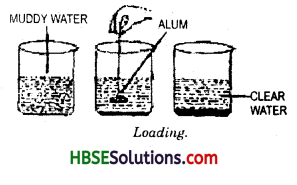
with a thread, is swirrled in such a water, then after some time the water becomes clear on standing. (Fig 5.7) The water becomes clear because the mud particles get loaded by the alum particles and become heavy and settle down at the bottom of the container and water can be decanted for further use.
![]()
12. Filtration : Filtration is used to separate solid impurities from a liquid. In our homes, tea is filtered to separate tea leaves and shikanji (lemo-nade) is filtered to separate the seeds of the lemon.
13. Evaporation : The process of separating any salt from its solution by removing water is called evaporation. This process is largely used to obtain common salt from sea water. When sea water is allowed to stand in shallow pits, water slowly turns into water vapours by absorbing the heat of the sun, leaving behind solid salt.
14. Magnetic separation : This process of separation is used when one of the components in the mixture is iron. For example a mixture of iron fillings and sand can 5.10. “Magnetic, separation”

be separated by moving a magnet in the mixture. The Magnet picks out only iron fillings from the sand. This process is called magnetic separation (Fig 5.10)
15. Churning: This process is used to separate cream from milk. When milk is churned for some time, the lighter cream particles float on its surface from where they can be separated. Churning can be done by hand or by using a mixi. (Fig 5.11.)
16. When one method of separation is not sufficient to separate the different substances present in a mixture, we use more than one methods. (See Fig 5.13) For example if we have a mixture of salt, water and sand and we want to separate all the three substances, we use two methods-decantation and evaporation.